vol. 24, No. 8, 2013, p. 1213-1384
Journal Impact Factor: Expectations and Hopes
In the modern digital era, June is a month of anxiety for everybody involved with publishing in sciences. June is the moment for divulgation by the Journal Citation Reports of the new impact mfactors and certainly we want to fly to the blue skies. High expectations and hopes may be a source of motivation, but they may act as a nest for frustration and disillusion.
It is important to have quantitative data in science and frequently Lord Kelvin is quoted by stating that "I often say that when you can measure what you are speaking about, and express it in numbers, you know something about it; but when you cannot express it in numbers, your knowledge is of a meager and unsatisfactory kind; it may be the beginning of knowledge, but you have scarcely, in your thoughts, advanced to the stage of science, whatever the matter may be".
(Read more at Editorial)
Nano-Assembled Supramolecular Films from Chitosan-Stabilized Gold Nanoparticles and Cobalt(II) Phthalocyanine
Anna T. B. Silva; Andreane G. Coelho; Lourdes C. da S. Lopes; Marccus V. A. Martins; Frank N. Crespilho; Arben Merkoçi; Welter C. da Silva
How to cite this article
How do gold nanoparticles can contributes to improve the charge transfer on supramolecular films? This paper shows that the nanoparticles effectively influence the kinetics of charge transfer in cobalt (II) phthalocyanine on a self-assembled surface. Details are presented in the Article Nano-Assembled Supramolecular Films from Chitosan-Stabilized Gold Nanoparticles and Cobalt(II) Phthalocyanine by Anna T. B.Silva, Andreane G. Coelho, Lourdes C. da S. Lopes, Marccus V. A. Martins, Frank N. Crespilho, Arben Merkoçi and Welter C. da Silva on page 1237.
https://dx.doi.org/10.5935/0103-5053.20130157
Editorial J. Braz. Chem. Soc. 2013, 24(8), 1213-1216
Journal Impact Factor: Expectations and Hopes
Joaquim A. Nóbrega; Watson Loh
How to cite this article
https://dx.doi.org/10.5935/0103-5053.20130175
Articles J. Braz. Chem. Soc. 2013, 24(8), 1217-1227
Simultaneous Determination of the Organochlorine and Pyrethroid Pesticides in Drinking Water by Single Drop Microextraction and Gas Chromatography
Elenice A. Carlos; Renata D. Alves; Maria Eliana L. R. de Queiroz; Antônio A. Neves
How to cite this article
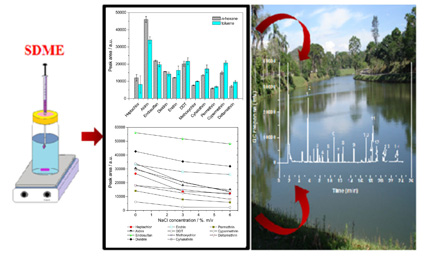
Single drop microextraction was proposed to determine fourteen pesticides in water samples. The method has demonstrated to be accurate, precise, rapid, simple and to require smaller volumes of organic solvent. The proposed method may be successfully applied for the drinking water monitoring.
https://dx.doi.org/10.5935/0103-5053.20130155
J. Braz. Chem. Soc. 2013, 24(8), 1228-1236
Tryptophan Determination at Carbon Fiber Ultramicroelectrodes by Fast-Scan Cyclic Voltammetry
Antônio R. Fiorucci; Priscila Cervini; Orlando Fatibello Filho; Éder T. G. Cavalheiro
How to cite this article
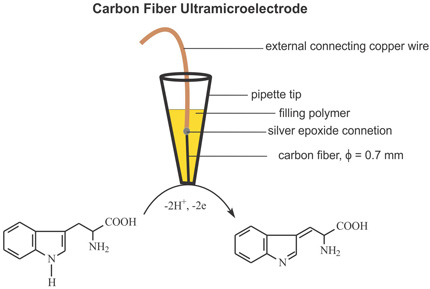
Electrochemical oxidation of tryptophan at a carbon fiber ultramicroelectrode.
https://dx.doi.org/10.5935/0103-5053.20130156
J. Braz. Chem. Soc. 2013, 24(8), 1237-1245
Nano-Assembled Supramolecular Films from Chitosan-Stabilized Gold Nanoparticles and Cobalt(II) Phthalocyanine
Anna T. B. Silva; Andreane G. Coelho; Lourdes C. da S. Lopes; Marccus V. A. Martins; Frank N. Crespilho; Arben Merkoçi; Welter C. da Silva
How to cite this article
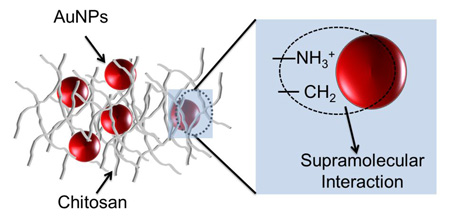
The synthesis and electrochemical properties of gold nanoparticles stabilized with chitosan, as well as their immobilization with cobalt(II) phthalocyanine to form layer-by-layer films, are reported.
https://dx.doi.org/10.5935/0103-5053.20130157
J. Braz. Chem. Soc. 2013, 24(8), 1246-1258
Oxide Film Growth by CSV on AISI 316L: a Combined Electrochemical and Analytical Characterization
Belén Díaz; Lorena Freire; Maria Fátima Montemor; Xosé Ramón Nóvoa
How to cite this article
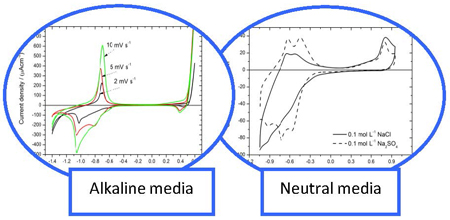
Oxide films formed in alkaline media on AISI 316L using cyclic sweep voltammetry (CSV) during the faster cycling rate (10 mV s-1). The higher scan rate promoted the formation of thinner films richer in Fe3+ species, mainly hydroxides, and more resistive. Neutral pH promoted a lower magnetite formation and more resistive layers with Mo presence.
https://dx.doi.org/10.5935/0103-5053.20130158
J. Braz. Chem. Soc. 2013, 24(8), 1259-1266
Electrochemical Degradation of Diuron in Chloride Medium using DSA® Based Anodes
Angelo R. F. Pipi; Sidney Aquino Neto; Adalgisa R. De Andrade
How to cite this article
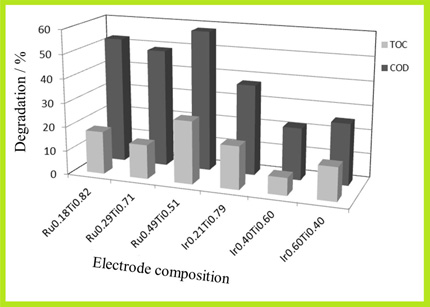
In this work, the degradation of diuron using electrodes of different compositions of ruthenium and iridium oxides was investigated. Ruthenium based anodes showed a better efficiency than iridium ones to remove the herbicide.
https://dx.doi.org/10.5935/0103-5053.20130159
J. Braz. Chem. Soc. 2013, 24(8), 1267-1275
Determination of Selenium, Chromium and Copper in Food Dyes by GF AAS
Emanueli do Nascimento da Silva; Nivaldo Baccan; Solange Cadore
How to cite this article
The manuscript describes the optimization and application of a method for determination of Se, Cr and Cu in food dyes using graphite furnace atomic absorption spectrometry.
https://dx.doi.org/10.5935/0103-5053.20130161
J. Braz. Chem. Soc. 2013, 24(8), 1276-1286
Redox Pathways of Aliskiren Based on Experimental and Computational Approach and Its Voltammetric Determination
Deniz Pamuk; İbrahim Hüdai Taşdemir; Abdulilah Ece; Esin Canel; Esma Kılıç
How to cite this article
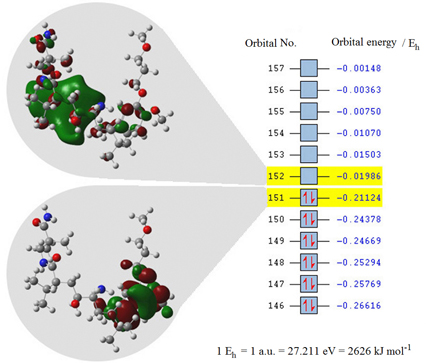
Aliskiren is proposed to be oxidized at the benzylic carbon and reduced at the amide carbonyl groups. Limits of detection and of quantification for aliskiren were found in the low μg L-1 range for the proposed methods.
https://dx.doi.org/10.5935/0103-5053.20130162
J. Braz. Chem. Soc. 2013, 24(8), 1287-1294
Sediment Removal from Crude Oil Emulsion using Microwave Radiation
Fabiane G. Antes; Juliana S. F. Pereira; Liange O. Diehl; Letícia S. F. Pereira; Paula Boeck; Regina C. L. Guimarães; Ricardo A. Guarnieri; Bianca M. S. Ferreira; Maria de Fatima P. dos Santos; Edson Luiz Foletto; Erico M. M. Flores
How to cite this article

Microwave radiation was applied to closed vessels for sediment removal. After irradiation, crude oil phase could be characterized due to low content of sediments, salt and water.
https://dx.doi.org/10.5935/0103-5053.20130163
J. Braz. Chem. Soc. 2013, 24(8), 1295-1306
New 1-Hydroxy-1,1-bisphosphonates Derived from 1H-Pyrazolo[3,4-b]pyridine: Synthesis and Characterization
Fátima C. Teixeira; Carla Lucas; M. João M. Curto; M. Neves; M. Teresa Duarte; Vânia André; António P. S. Teixeira
How to cite this article
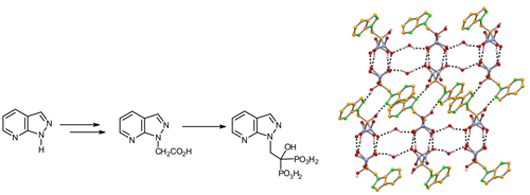
The first synthesis of phosphonate derived from pyrazolo[3,4-b]pyridine was achieved by a one-pot strategy from their corresponding carboxylic acids. An X-ray structure of one bisphosphonate is presented.
https://dx.doi.org/10.5935/0103-5053.20130164
J. Braz. Chem. Soc. 2013, 24(8), 1307-1313
Kinetics and Mechanism of Oxidation of the Drug Intermediate 1-(2-Hydroxyethyl)piperidine by Bis(hydrogenperiodato)argentate(III)
Hongmei Shi; Jiong Zhang; Shuying Huo; Shigang Shen; Weijun Kang; Tiesheng Shi
How to cite this article
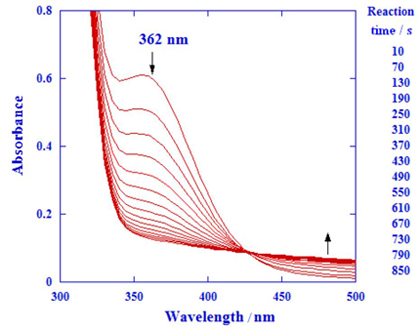
The oxidation of the drug intermediate 1-(2-hydroxyethyl)piperidine by [Ag(HIO6)2]5- was carefully studied by spectroscopic techniques, leading to the determination of the rate constants and associated activation parameters. A consistent reaction mechanism was proposed from the derived rate law allowing the in depth understanding of that reaction.
https://dx.doi.org/10.5935/0103-5053.20130165
J. Braz. Chem. Soc. 2013, 24(8), 1314-1321
Clusiaxanthone and Tocotrienol Series from Clusia pernambucensis and their Antileishmanial Activity
Everton M. Silva; Renata M. Araújo; Lindomar G. Freire-Filha; Edilberto R. Silveira; Norberto P. Lopes; José Elias de Paula; Raimundo Braz-Filho; Laila S. Espindola
How to cite this article
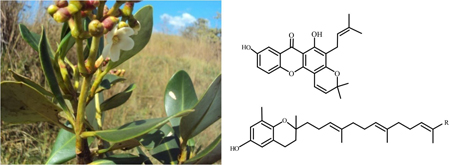
This work describes the isolation of a previously unreported xanthone and a series of tocotrienols from Clusia pernambucensis and their activity against Leishmania (Leishmania) amazonensis.
https://dx.doi.org/10.5935/0103-5053.20130166
J. Braz. Chem. Soc. 2013, 24(8), 1322-1328
Influence of the Structure on the Antioxidant Activity of Tetradentate Schiff Bases and their Copper(II) Complexes: Possible Mechanisms
Najat M. Aburas; Nikola R. Stevanović; Miloš K. Milčić; Aleksandar Đ. Lolić; Maja M. Natić; Zivoslav Lj. Tešić; Rada M. Baošić
How to cite this article
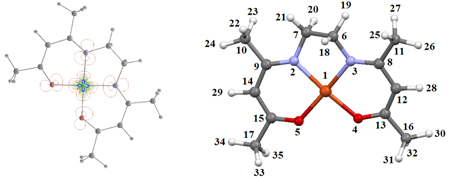
A total of 5 tetradentate Schiff bases and their copper(II) complexes were tested for antioxidant activity. Descriptors calculated by hybrid DFT method provided insight into their antioxidant behavior and mechanism of the antioxidant activity.
https://dx.doi.org/10.5935/0103-5053.20130167
J. Braz. Chem. Soc. 2013, 24(8), 1329-1338
Synthesis of Flower-like Ag-ZnO Nanostructure and its Application in the Photodegradation of Methyl Orange
Mansour Arab Chamjangali; Samira Boroumand
How to cite this article

Flower-like Ag-ZnO was more efficient than ZnO as photocatalyst for degradation of methyl orange. The possible factors influencing the photocatalytic activity were tested and discussed in order to find the best conditions.
https://dx.doi.org/10.5935/0103-5053.20130168
J. Braz. Chem. Soc. 2013, 24(8), 1339-1344
Electrochemical Hydrogen-Storage Properties of La0.78Mg0.22Ni2.67Mn0.11Al0.11Co0.52-M1Ni3.5Co0.6Mn0.4Al0.5 Composites
Hongxia Huang; Guohui Li; Shuxin Zhuang
How to cite this article
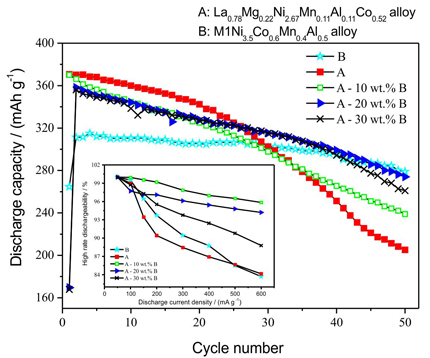
With the addition of M1Ni3.5Co0.6Mn0.4Al0.5 (M1 denotes mischmetal) alloy, the cycle stability of AB3-type La0.78Mg0.22Ni2.67Mn0.11Al0.11Co0.52 hydrogen storage alloy electrode is significantly improved. Moreover, the electrochemical kinetics increases first and then decreases with the increase of wt.% B.
https://dx.doi.org/10.5935/0103-5053.20130169
J. Braz. Chem. Soc. 2013, 24(8), 1345-1350
Metal Corrosion in Polychrome Baroque Lead Sculptures: a Case Study
Dalva L. A. de Faria; Thiago S. Puglieri; Luiz Antonio C. Souza
How to cite this article
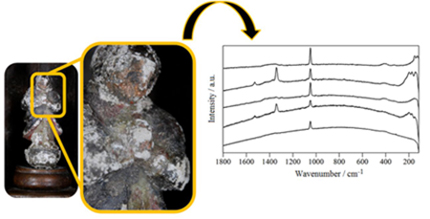
Corroded polychrome lead sculptures from Oratory Museum (Ouro Preto, Minas Gerais, Brazil) were analyzed by different techniques. Environmental simulations supported that the degradation process was caused mainly by the curing process of the painted showcase baseplate, although cleaning products proved to be potentially harmful to Pb objects.
https://dx.doi.org/10.5935/0103-5053.20130170
J. Braz. Chem. Soc. 2013, 24(8), 1351-1356
Prediction of Parameters (Soluble Solid and pH) in Intact Plum using NIR Spectroscopy and Wavelength Selection
Rosangela C. Costa; Kássio M. G. de Lima
How to cite this article
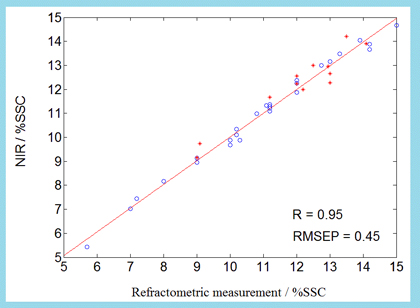
In this work, near-infrared reflectance spectroscopy and multivariate calibration for prediction of parameters (soluble solid and pH) were used in intact plum.
https://dx.doi.org/10.5935/0103-5053.20130172
J. Braz. Chem. Soc. 2013, 24(8), 1357-1365
Basis Set Convergence on Static Electric Dipole Polarizability Calculations of Alkali-Metal Clusters
Fábio A. L. de Souza; Francisco E. Jorge
How to cite this article
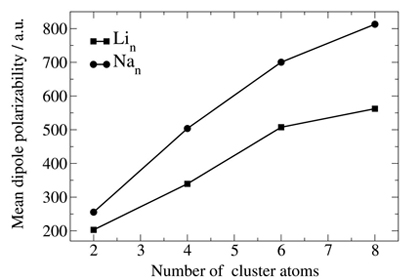
Estimated CBS-MP2 mean dipole polarizability vs. cluster size.
https://dx.doi.org/10.5935/0103-5053.20130171
J. Braz. Chem. Soc. 2013, 24(8), 1366-1372
Modeling Controlled Potassium Release from Phlogopite in Solution: Exploring the Viability of using Crushed Phlogopitite Rock as an Alternative Potassium Source in Brazilian Soil
Adriana de A. S. da Silva; João A. Sampaio; Adão B. da Luz; Sílvia C. A. França; Célia M. Ronconi
How to cite this article
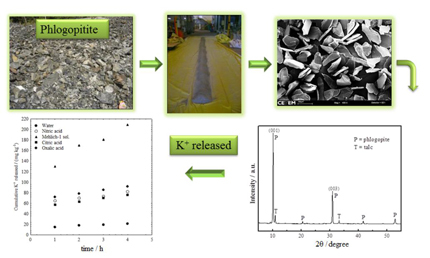
Phlogopitite is a rock that is disposed of as a byproduct of emerald mining in Brazil. It contains the mineral phlogopite, rich in potassium. Kinetic studies on potassium release in solution were conducted to evaluate this rock as an alternative potassium source for crops.
https://dx.doi.org/10.5935/0103-5053.20130173
J. Braz. Chem. Soc. 2013, 24(8), 1373-1379
Evaluation of the Oxidative Stability of Biodiesel Blends from Soybean, Tallow and Castor Bean using Experimental Mixture Design
Anaildes L. Carvalho; Sarah M. F. Santana; Cristiane S. Silva; Iuri M. Pepe; Marcos A. Bezerra; Leandro M. Aragão; Cristina M. Quintella; Leonardo S. G. Teixeira
How to cite this article
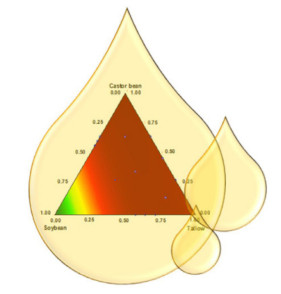
An experimental design of biodiesel constrained mixtures was employed to evaluate and optimize the oxidative stability of soybean biodiesel mixed with different amounts of tallow and castor bean biodiesels.
https://dx.doi.org/10.5935/0103-5053.20130174
Short Report J. Braz. Chem. Soc. 2013, 24(8), 1380-1384
Thermometric Quantitative Selective Analysis of Sodium Methoxide in Methanol Industrial Solutions
Flavio A. Bastos; Sabir Khan; Eduardo H. Simões; Carlos A. Teixeira; Matthieu Tubino
How to cite this article
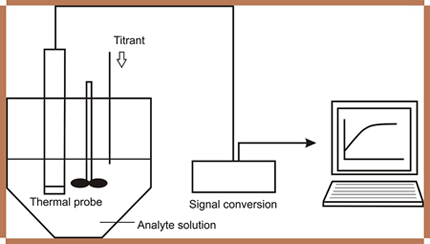
A solution of pure water at 10% (v v-1) in methanol was used as the thermometric reagent (titrant) for the quantitation of metoxide in methanol solution. The temperature variation measurements were performed essentially using a thermistor. The data were collected and analyzed graphically in a computer.
https://dx.doi.org/10.5935/0103-5053.20130160
Online version ISSN 1678-4790 Printed version ISSN 0103-5053
Journal of the Brazilian Chemical Society
JBCS Editorial and Publishing Office
University of Campinas - UNICAMP
13083-970 Campinas-SP, Brazil
Free access











