vol. 22, No. 4, 2011, p. 605-807
The Brazilian Chemical Industry - Challenges and Opportunities
Chemistry is a key sector in any economy, with a strong presence in practically all productive chains. It is no coincidence that the major world economies are also the leaders in the manufacturing of chemicals. Many countries have developed, created several employment opportunities, added value to their industries out of investments that started with the expansion in the production of chemicals.
(Read more at Editorial)
Editorial J. Braz. Chem. Soc. 2011, 22(4), 605-608
Review J. Braz. Chem. Soc. 2011, 22(4), 609-622
Comprehensive Two-Dimensional Gas Chromatography for Analysis of Volatile Compounds in Foods and Beverages
Juliane Elisa Welke; Cláudia Alcaraz Zini
How to cite this article
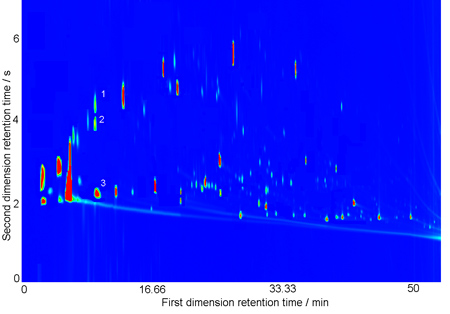
GC x GC color plot of a Brazilian base wine obtained with a polar and a medium-polar column set.
https://dx.doi.org/10.1590/S0103-50532011000400002
Articles J. Braz. Chem. Soc. 2011, 22(4), 623-636
Metal Halide Hydrates as Lewis Acid Catalysts for the Conjugated Friedel-Crafts Reactions of Indoles and Activated Olefins
Cristiane S. Schwalm; Marco Antonio Ceschi; Dennis Russowsky
How to cite this article
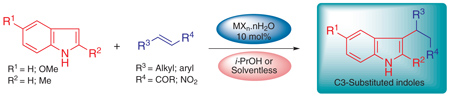
Metal halide hydrates are efficient catalysts to the Conjugate Friedel-Crafts reaction (CFC) of Indoles and activated olefins in alcoholic media. Solvent-free conditions were successful applied to prepare nitro-adducts and b-carbolines in good yields in an environmentally-friend protocol.
https://dx.doi.org/10.1590/S0103-50532011000400003
J. Braz. Chem. Soc. 2011, 22(4), 637-642
2D Chemical Drawings Correlate to Bioactivities: MIA-QSAR Modelling of Antimalarial Activities of 2,5-Diaminobenzophenone Derivatives
Rodrigo A. Cormanich; Matheus P. Freitas; Roberto Rittner
How to cite this article
QSAR based on 2D chemical images (MIA-QSAR) shows that bidimensional structures correlate with biological activities, and is a promising tool for the design of new antimalarial compounds, with modeling capability comparable to well established 3D approaches.
https://dx.doi.org/10.1590/S0103-50532011000400004
J. Braz. Chem. Soc. 2011, 22(4), 643-647
Quantification of Essential Fatty Acids in the Heads of Nile Tilapia (Oreochromis niloticus) Fed with Linseed Oil
Ana C. Aguiar; Solange M. Cottica; Marcela Boroski; Cláudio C. Oliveira; Elton G. Bonafé; Polyana B. França; Nilson E. Souza; Jesui V. Visentainer
How to cite this article
The fatty acid methyl esters (FAME) were prepared by methylation of the total lipids (TL) and separated by gas chromatography using as parameter the equivalent chain-length values (ECL) in the identification and flame Ionization detector response factors in the quantification.
https://dx.doi.org/10.1590/S0103-50532011000400005
J. Braz. Chem. Soc. 2011, 22(4), 648-651
Determination of p-Phenylenediamine by Perturbation of a Non-Equilibrium Stationary State in the B-Z Reaction
Jinzhang Gao; Xiaodong Chen Jie Ren; Wu Yang
How to cite this article
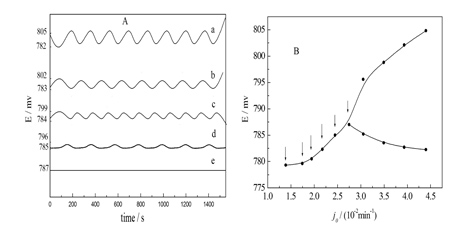
Figure A shows a regular oscillator being translating gradually into a non-equilibrium stationary state. Figure B shows a bifurcation point.
https://dx.doi.org/10.1590/S0103-50532011000400006
J. Braz. Chem. Soc. 2011, 22(4), 652-659
Electrochemical Feasibility Study of Methyl Parathion Determination on Graphite-Modified Basal Plane Pyrolytic Graphite Electrode
Leonardo Luiz Okumura; Adelir Aparecida Saczk; Marcelo Firmino de Oliveira; Ana Carolina C. Fulgêncio; Larissa Torrezani; Paulo Eduardo N. Gomes; Rosana M. Peixoto
How to cite this article
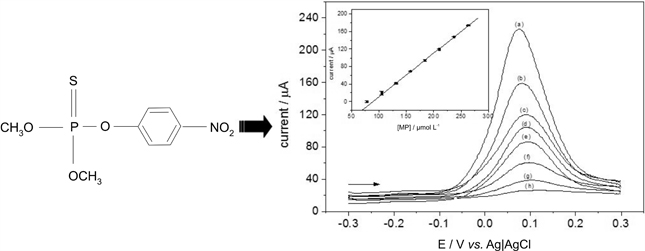
SWV optimized responses for different MP concentrations in supporting electrolyte 0.20 mol L-1 acetate buffer pH 5.2 and volume of graphite suspension as a modifier of 10 mL. Inset: calibration curve for MP.
https://dx.doi.org/10.1590/S0103-50532011000400007
J. Braz. Chem. Soc. 2011, 22(4), 660-668
Synthesis, Characterization and Catalytic Activity of Two Novel cis-Dioxovanadium(V) Complexes: [VO2(L)] and [VO2(HLox)]
Natália M. L. Silva; Carlos B. Pinheiro; Eluzir P. Chacon; Jackson A. L. C. Resende; José Walkimar de M. Carneiro; Tatiana L. Fernández; Marciela Scarpellini; Mauricio Lanznaster
How to cite this article
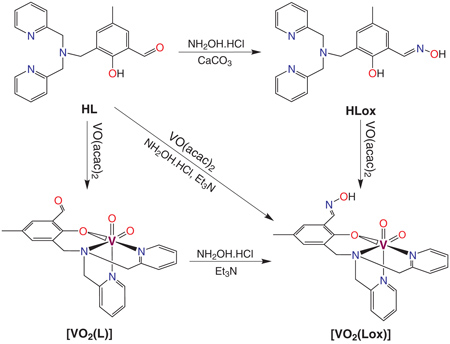
The complexes [VO2(L)] and [VO2(HLox)] were synthesized by the reaction of [VO(acac)2] with the ligands HL and H2Lox, respectively. Alternatively, [VO2(HLox)] was also obtained by the reaction of HL with [VO(acac)2] and hydroxylamine, or reacting [VO2(L)] with hydroxylamine.
https://dx.doi.org/10.1590/S0103-50532011000400008
J. Braz. Chem. Soc. 2011, 22(4), 669-676
EPR and Magnetic Studies of a Carboxylate-Bridged Dinuclear Copper(II) Compound: [Cu2(flu)4(dmf)2]
Otaciro R. Nascimento; Lia M. B. Napolitano; María H. Torre; Octavio Peña; Rafael Calvo
How to cite this article
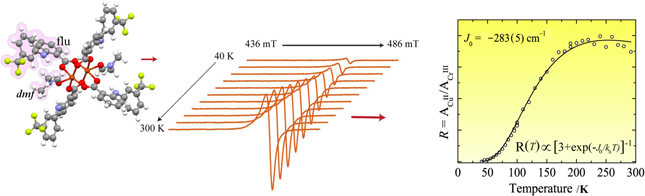
In the GA we are showing the EPR line variation with temperature of single-crystal [Cu2(flu)4(dmf)2] compound and magnetic susceptibility versus temperature extracted from this measurements.
https://dx.doi.org/10.1590/S0103-50532011000400009
J. Braz. Chem. Soc. 2011, 22(4), 677-683
Pre-concentration of Lithium Prior to Its Determination in Honey by Flame Optical Emission Spectrometry
Helena Stecka; Pawel Pohl
How to cite this article
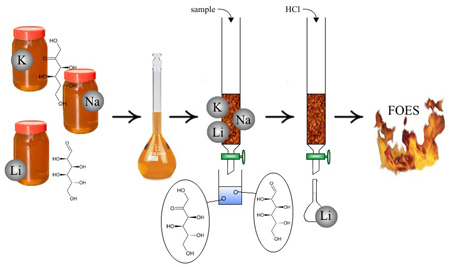
A procedure based on solid phase extraction with a strongly acidic cation-exchanger Dowex 50Wx
8-200 was proposed to pre-concentrate and determine ultra-traces of Li in floral honeys. It enabled to determine from 0.4 to 5.3 ng g-1 of Li with precision being within 8-11%.
https://dx.doi.org/10.1590/S0103-50532011000400010
J. Braz. Chem. Soc. 2011, 22(4), 684-692
QSPR Modeling using Catalan Solvent and Solute Parameters
Abolghasem Jouyban; Mohammad A. A. Fakhree; Ali Shayanfar; Taravat Ghafourian
How to cite this article
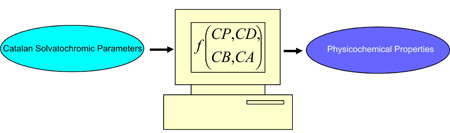
The Catalan solvent parameters were used to develop a new set of solute parameters. The developed solute parameters were used to propose a novel model for predicting aqueous solubility of solutes. The Catalan solvent parameters were employed to develop models for representing a number of physico-chemical properties of the solvents.
https://dx.doi.org/10.1590/S0103-50532011000400011
J. Braz. Chem. Soc. 2011, 22(4), 693-701
Electrochemical Oxidation of Caffeic and Ferulic Acid Derivatives in Aprotic Medium
Magali Salas-Reyes; Javier Hernández; Zaira Domínguez; Felipe J. González; Pablo D. Astudillo; Rosa Elena Navarro; Evelin Martínez-Benavidez; Carlos Velázquez-Contreras; Samuel Cruz-Sánchez
How to cite this article
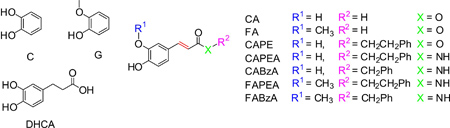
The electrochemical oxidation of a series of caffeic and ferulic acids derivatives as well as their corresponding redox moieties catechol and guaiacol was studied as a function of the structure in aprotic medium.
https://dx.doi.org/10.1590/S0103-50532011000400012
J. Braz. Chem. Soc. 2011, 22(4), 702-708
Binding Events of (S)-N-(3-Oxo-octanoyl)-homoserine Lactone with Agrobacterium tumefaciens Mutant Cells Studied by Saturation Transfer Difference NMR
Luís Fernando Cabeça; Armando Mateus Pomini; Pedro Luiz R. Cruz; Anita J. Marsaioli
How to cite this article
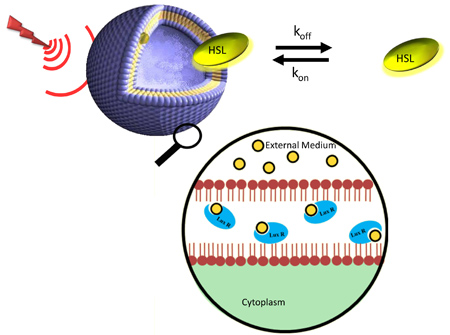
Upper Figure: Saturation of a liposome 1H NMR signal transferred to the complex liposome-homoserine lactone (HSL)and dissociation produce free homoserine lactone with saturated hydrogens. Lower Figure: First binding events of the quorum sensing phenomenum showing the HSL diffusing through the bacterium membrane.
https://dx.doi.org/10.1590/S0103-50532011000400013
J. Braz. Chem. Soc. 2011, 22(4), 709-717
Application of Pyrite and Chalcopyrite Electrodes for the Acid-Base Determinations in Nitriles
Zoran Simić; Zorka D. Stanić; Milan Antonijević
How to cite this article
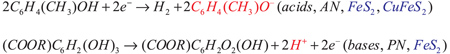
In the course of the coulometric determinations of acids (bases) in non-aqueous solvents (AN and PN), the electrochemical generation of lyate (hydrogen) ions is performed by the reduction (oxidation) of an appropriate electroreactive compounds. The natural minerals pyrite (FeS2) and chalcopyrite (CuFeS2) have been used as the indicator electrodes.
https://dx.doi.org/10.1590/S0103-50532011000400014
J. Braz. Chem. Soc. 2011, 22(4), 718-725
Nanoporous of W/WO3 Thin Film Electrode Grown by Electrochemical Anodization Applied in the Photoelectrocatalytic Oxidation of the Basic Red 51 used in Hair Dye
Luciano E. Fraga; Maria Valnice B. Zanoni
How to cite this article
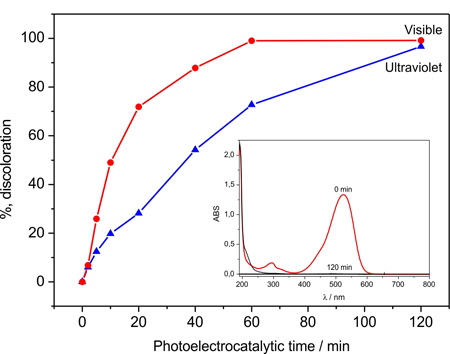
This image shows UV-Vis spectra and the present total color removal after 120 min of the 1.0 x10-5 mol L-1 basic red solution when treated by photoelectrocatalytic oxidation using both systems of irradiation (visible and ultraviolet) obtained before and after 120 min.
https://dx.doi.org/10.1590/S0103-50532011000400015
J. Braz. Chem. Soc. 2011, 22(4), 726-735
A Low-Cost Portable Microcontrolled Nephelometer for Potassium Determination
Vagner B. dos Santos; Thiago B. Guerreiro; Willian T. Suarez; Ronaldo C. Faria; Orlando Fatibello-Filho
How to cite this article
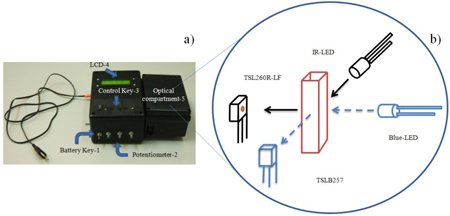
Low-cost portable microcontrolled nephelometer. a) Picture of the nephelometer developed; b) Schematic diagram of the optical compartment showing the blue LED and photodiode TSLB257 at 90º and infrared LED and TSL260R-LF at 90º.
https://dx.doi.org/10.1590/S0103-50532011000400016
J. Braz. Chem. Soc. 2011, 22(4), 736-740
Visible LED-Based Instrumentation for Photometric Determination of Electroosmotic Flow in Microchannels
José Alberto Fracassi da Silva; Ariane Deblire; Dosil P. de Jesus; Wendell Karlos T. Coltro
How to cite this article
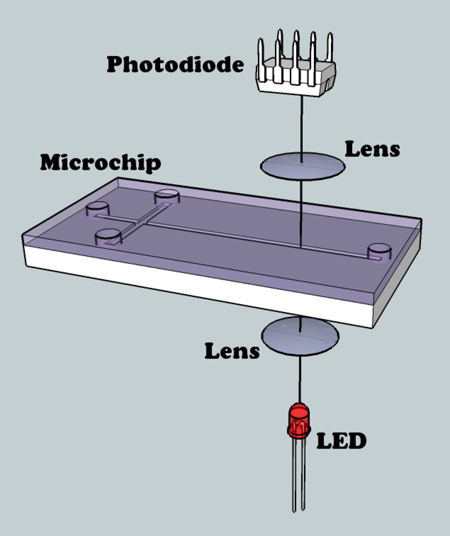
In this article we demonstrate how to use an inexpensive instrumentation based on visible-high intensity LED and microscope for the determination of the electroosmotic flow in microseparation devices.
https://dx.doi.org/10.1590/S0103-50532011000400017
J. Braz. Chem. Soc. 2011, 22(4), 741-745
Chemical Analysis of Essential Oils from Ocotea gomezii W.C. Burger and Ocotea morae Gómez-Laur. (Lauraceae) Collected at
Carlos Chaverri; Cecilia Díaz; José F. Cicció
How to cite this article
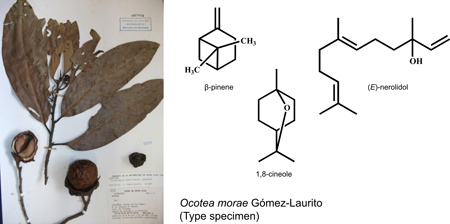
Morphology of Ocotea morae (Lauraceae) and structural formulas of major constituents from leaves (b-pinene), bark (1,8-cineole) and wood [(E)-nerolidol] essential oils.
https://dx.doi.org/10.1590/S0103-50532011000400018
J. Braz. Chem. Soc. 2011, 22(4), 746-755
Development of Nano Poly(3-methyl thiophene)/Multiwalled Carbon Nanotubes Sensor for the Efficient Detection of Some Pesticides
Palaniappan Abirama Sundari; Paramasivam Manisankar
How to cite this article
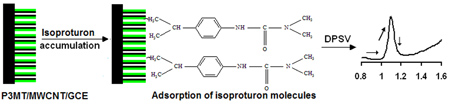
We demonstrate the fabrication of nanosize poly(3-methyl thiophene) (P3MT)/ multiwalled carbon nanotubes (MWCNTs) modified glassy carbon (GCE) electrode for the sensitive determination of some common pesticides.
https://dx.doi.org/10.1590/S0103-50532011000400019
J. Braz. Chem. Soc. 2011, 22(4), 756-766
An Alternative Mechanism for the 1,4-Asymmetric Induction in the Stereoselective Addition of (R)-Pantolactone to 2-Phenylpropylketene
Alexander M. Silva; Clarissa O. da Silva; André G. H. Barbosa; Rosane A. Fontes; Sergio Pinheiro; Marco Edilson F. Lima; Rosane N. Castro
How to cite this article
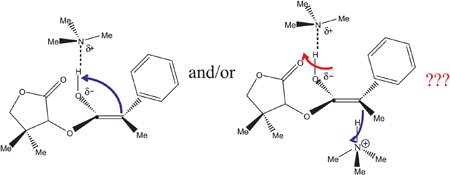
Two possible mechanisms for the 1,4-asymmetric induction in the stereoselective addition of (R)-pantolactone to 2-phenylpropylketene.
https://dx.doi.org/10.1590/S0103-50532011000400020
J. Braz. Chem. Soc. 2011, 22(4), 767-771
Electrochemical Behavior and Determination of Fluconazole
Éric de S. Gil; Diogo D. Cordeiro; Ana E. B. Matias; Sílvia H. P. Serrano
How to cite this article
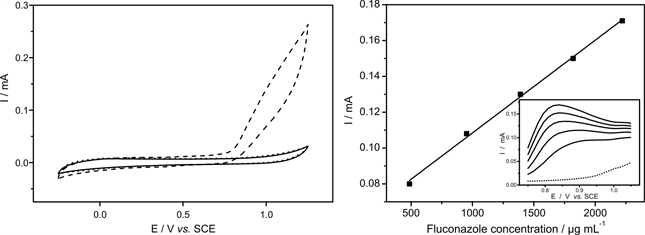
Voltammograms and the analytical curve obtained at glassy carbon electrode in 0.1 mol L-1 phosphate buffer solution, pH 8.0 obtained at glassy carbon electrode for blank and fluconazole solutions.
https://dx.doi.org/10.1590/S0103-50532011000400021
J. Braz. Chem. Soc. 2011, 22(4), 772-779
Nutrients in Amazonian Black Earth from Caxiuanã Region
Vanda P. Lemos; Antônio R. de Oliveira Meireles; Kelly das Graças Fernandes; Milena C. de Moraes; Marcondes L. da Costa; Any K. Terra Silva; Dirse C. Kern
How to cite this article
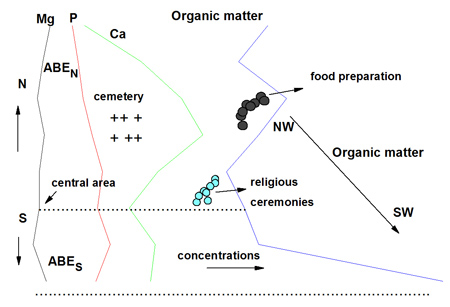
Trend of the concentrations of Mg, Ca, P and organic matter in archaeological black earth (ABE) at the site Ilha de Terra from Caxiuana suggests placeholders as cemetery, religious ceremonies and food preparations by ancient inhabitants of this site.
https://dx.doi.org/10.1590/S0103-50532011000400022
J. Braz. Chem. Soc. 2011, 22(4), 780-789
Synthesis, Characterization and Photovoltaic Properties of Di-Anchoring Organic Dyes
Tzi-Yi Wu; Ming-Hsiu Tsao; Shyh-Gang Su; H. Paul Wang; Yuan-Chung Lin; Fu-Lin Chen; Cheng-Wen Chang; I-Wen Sun
How to cite this article
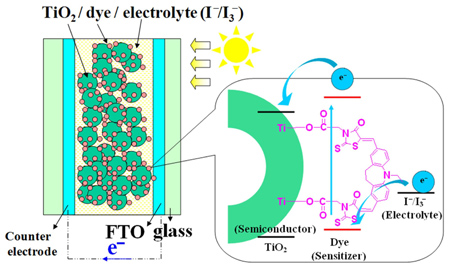
Organic dyes comprising carbazole, iminodibenzyl and phenothiazine moieties, respectively, as the electron donors and di-anchoring rhodanine rings as the electron acceptors are synthesized and evaluated for use in dye-sensitized solar cells. Solar cells employing these organic dyes show good overall conversion efficiency (up to 4.91%).
https://dx.doi.org/10.1590/S0103-50532011000400023
Short Reports J. Braz. Chem. Soc. 2011, 22(4), 790-795
Exhausted Yerba Mate Leaves (Ilex paraguariensis) as Biosorbent for the Removal of Metals from Aqueous Solutions
Guillermo J. Copello; Rodolfo E. Garibotti; Florencia Varela; María V. Tuttolomondo; Luis E. Diaz
How to cite this article

Exhausted yerba mate leaves (Ilex paraguariensis) demonstrated to effectively adsorb metals from aqueous solutions, such as Cr(VI), which suggest the applicability of this cost effective biosorbent.
https://dx.doi.org/10.1590/S0103-50532011000400024
J. Braz. Chem. Soc. 2011, 22(4), 796-800
New Adducts of Lapachol with Primary Amines
Mirelly D. F. Santos; José T. Litivack-Junior; Roberto V. Antunes; Tania M. S. Silva; Celso A. Camara
How to cite this article
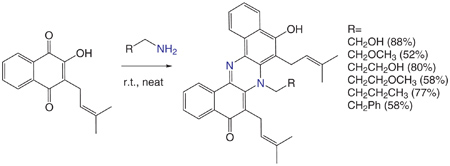
In this reaction substituted phenazines are formed by the neat reaction of alkylamines with lapachol, and in the figure the nitrogens are painted in blue to emphazise that one of the ring is originated from a Strecker degradation reaction.
https://dx.doi.org/10.1590/S0103-50532011000400025
J. Braz. Chem. Soc. 2011, 22(4), 801-806
Fast Analysis of Taurine in Energetic Drinks by Electrospray Ionization Mass Spectrometry
Rodrigo R. Catharino; Renato Haddad; Helena T. Godoy; Marcos N. Eberlin; Leonardo S. Santos
How to cite this article
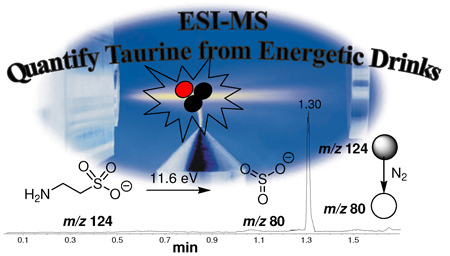
Direct infusion ESI-MS/MS in the negative ion mode using SRM (m/z 124 -> m/z 80) was applied with high confidence, speed and selectivity to quantifty taurine in energetic drinks.
https://dx.doi.org/10.1590/S0103-50532011000400026
Additions and Corrections J. Braz. Chem. Soc. 2011, 22(4), 807
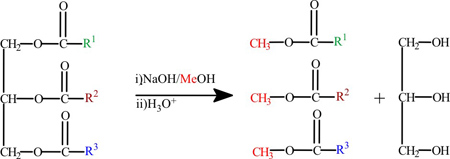
Evaluation of Snake Venom Phospholipase A2: hydrolysis of Non-Natural Esters
Renan A. S. Pirolla; Paulo A. Baldasso; Sérgio Marangoni; Paulo J. S. Moran; José Augusto R. Rodrigues
How to cite this article
https://dx.doi.org/10.1590/S0103-50532011000400027
Online version ISSN 1678-4790 Printed version ISSN 0103-5053
Journal of the Brazilian Chemical Society
JBCS Editorial and Publishing Office
University of Campinas - UNICAMP
13083-970 Campinas-SP, Brazil
Free access










