vol. 23, No. 6, 2012, p. 985-1198
High School Chemistry Teaching: How to Improve it?
On its 35th anniversary, the Brazilian Chemical Society (BCS) reaffirms itself as one of the most prominent scientific societies in Brazil. Its importance for the high standard reached by the Brazilian university system, since its foundation in 1977, can be measured by the several Presidents and Vice-Presidents of Universities from all over the country that have been part of the BCS Boards and Advisory Councils or who have been Regional Secretaries of the BCS. A number of these leaders were forged in the memorable general assemblies of the BCS Annual Meetings.
(Read more at Editorial)
Surface Properties of Oxidized and Aminated Multi-Walled Carbon Nanotubes
Wellington M. Silva; Hélio Ribeiro; Luciana M. Seara; Hállen D. R. Calado; André S. Ferlauto; Roberto M. Paniago; Cristiano F. Leite; Glaura G. Silva
How to cite this article
Functionalization or chemical modification of carbon nanotubes through the covalent attachment of atoms or molecules to the surface has been used to introduce chemical specificity allowing their processability in different environments. Evaluating the functionalization distribution of groups on the carbon nanotube surface is an important task. It can be achieved by monitoring the phase shift with electric force microscopy (EFM). The images obtained give qualitative information of the local charge distribution through contrast in areas where the conductivity varies.
https://dx.doi.org/10.1590/S0103-50532012000600012
Editorial J. Braz. Chem. Soc. 2012, 23(6), 985-986
Account J. Braz. Chem. Soc. 2012, 23(6), 987-1007
The Impressive Chemistry, Applications and Features of Ionic Liquids: Properties, Catalysis & Catalysts and Trends
Brenno A. D. Neto; John Spencer
How to cite this article
The chemistry of ionic liquids is impressive. No doubt! In this article it is overviewed some contributions to the development of catalysis and catalysts in ionic liquids, especially those with imidazolium-based derivatives. Due to the importance of ionic liquids, some physicochemical properties of interest and their supramolecular organization are also described
https://dx.doi.org/10.1590/S0103-50532012000600002
Articles J. Braz. Chem. Soc. 2012, 23(6), 1008-1017
Chemical Modification of Polyaniline by N-Grafting of Polystyrenic Chains Synthesized via Nitroxide-Mediated Polymerization
Maryam Hatamzadeh; Ali Mahyar; Mehdi Jaymand
How to cite this article
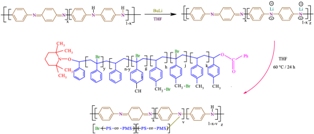
Conductive N-substituted polyaniline was synthesized by incorporation of brominated polystyrenic chains onto emeraldine form of polyaniline. The obtained terpolymer was characterized using FTIR, DSC, TGA, UV-Vis and cyclic voltammetry
https://dx.doi.org/10.1590/S0103-50532012000600003
J. Braz. Chem. Soc. 2012, 23(6), 1018-1023
Kinetic Modeling of the Alkaline Decomposition of Potassium Arsenojarosite
Mizraim U. Flores; Francisco Patiño; Iván A. Reyes; Isauro Rivera; Martín Reyes; Julio C. Juárez
How to cite this article

The kinetic model that describes the alkaline decomposition of potassium arsenojarosite in NaOH and Ca(OH)2 media was obtained and verified
https://dx.doi.org/10.1590/S0103-50532012000600004
J. Braz. Chem. Soc. 2012, 23(6), 1024-1032
The Influence of the Amount and Impregnation Sequence of Potassium on the Catalytic Performance of VOx/Al2O3 for the Non-Oxidative Dehydrogenation of n-Butane
María A. Volpe; Elba A. García
How to cite this article
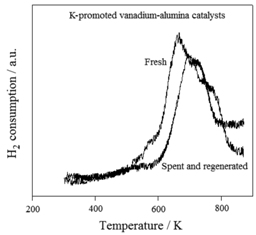
This figure shows TPR (temperature programmed reduction) profiles of fresh and regenerated K-promoted vanadium-alumina catalysts. The differences observed in these TPR patterns confirm that the deactivation of these catalysts may be related with irreversible structural changes in the vanadium oxide speciation
https://dx.doi.org/10.1590/S0103-50532012000600005
J. Braz. Chem. Soc. 2012, 23(6), 1033-1040
Dispersive Liquid-Liquid Microextraction Procedure for the Determination of Palladium by Flame Atomic Absorption Spectroscopy
Şerife Saçmaci; Şenol Kartal; Sevilay Dural
How to cite this article
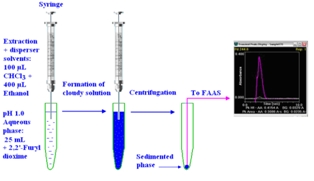
The important steps of dispersive liquid-liquid microextraction procedure for the determination of palladium(II) using 2,2'-furyldioxime. A new dispersive liquid-liquid microextraction system was developed for the determination of Pd(II) in catalytic converter, anode slime, road sediment and water samples. The compound 2,2'-furyldioxime as a complexing agent was firstly used in the determination of Pd(II) by flame atomic absorptin spectrometry using dispersive liquid-liquid microextraction procedure
https://dx.doi.org/10.1590/S0103-50532012000600006
J. Braz. Chem. Soc. 2012, 23(6), 1041-1047
Chemical Composition Similarity between the Essential Oils Isolated from Male and Female Specimens of Each Five Baccharis Species
Michele A. Besten; Vanessa C. G. Jasinski; Ângela de G. L. C. Costa; Domingos S. Nunes; Sávio L. Sens; Alberto Wisniewski Jr.; Edesio L. Simionatto; Dilamara Riva; Juliana B. Dalmarco; Daniel Granato
How to cite this article
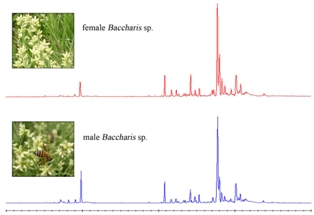
The analytical data presented in this study demonstrate that there may be a high degree of similarity between the compositions of essential oils from male and female specimens of Baccharis species
https://dx.doi.org/10.1590/S0103-50532012000600007
J. Braz. Chem. Soc. 2012, 23(6), 1048-1053
A Validated Chiral LC Method for the Enantiomeric Separation of Repaglinide on Immobilized Amylose Based Stationary Phase
Kiran Patil; Vipul Rane; Ravindra Yeole; Devanand Shinde
How to cite this article
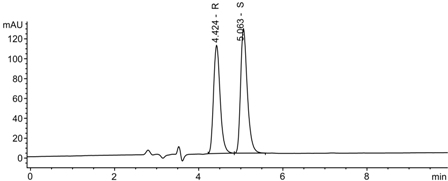
Enatiomeric resolution of repaglinide achieved on Chiralpak IA column using n-hexane:ethanol:TFA (80:20:0.2, v/v/v)
https://dx.doi.org/10.1590/S0103-50532012000600008
J. Braz. Chem. Soc. 2012, 23(6), 1054-1061
11H HRMAS NMR Spectroscopy and Chemometrics for Evaluation of Metabolic Changes in Citrus sinensis Caused by Xanthomonas axonopodis pv. citri
Lorena M. A. Silva; Elenilson G. Alves Filho; Rafael Choze; Luciano M. Lião; Glaucia B. Alcantara
How to cite this article
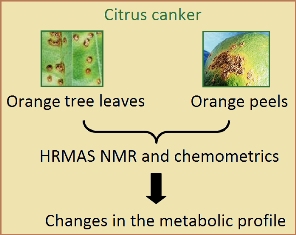
Orange tree leaves and orange peels attacked by citrus canker were analyzed by HRMAS NMR and chemometrics. Changes in metabolic profile have highlighted biochemical responses
https://dx.doi.org/10.1590/S0103-50532012000600009
J. Braz. Chem. Soc. 2012, 23(6), 1062-1069
An Efficient Synthesis of D-Galactose-Based Multivalent Neoglycoconjugates
Saulo F. de Andrade; Rute C. Figueiredo; José D. de Souza Filho; Ricardo J. Alves
How to cite this article
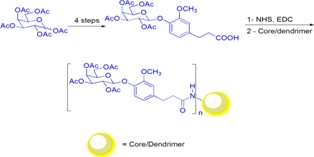
An efficient synthesis of five D-galactose-based multivalent neoglycoconjugates from peracetylated D-galactose is reported. Glycodendrimers were obtained in few steps using cheap reactants in 44-55% overall yields
https://dx.doi.org/10.1590/S0103-50532012000600010
J. Braz. Chem. Soc. 2012, 23(6), 1070-1077
Fabrication, Characterization and Analytical Performance of the Hydroxylamine Sensor Based on an Oracet Blue Multi-Walled Carbon Nanotubes Film Deposited on an Electrode Surface
Hamid R. Zare; Navid Nasirizadeh
How to cite this article
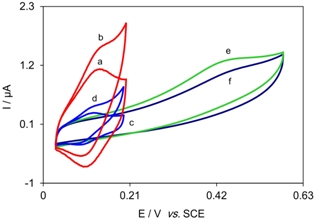
Cyclic voltammograms of a glassy carbon electrode modified with oracet blue and multi-walled carbon nanotubes (OBMWCNT-GCE) in a 0.1 mol L-1 phosphate buffer solution (pH 7.0) at scan rate 20 mV s-1 in (a) the absence and (b) the presence of 0.50 mmol L-1 hydroxylamine. (d), (e) and (f) as (b) for OBMGCE, MWCNT-GCE and activated GCE, respectively. (c) as (a) for OBMGCE
https://dx.doi.org/10.1590/S0103-50532012000600011
J. Braz. Chem. Soc. 2012, 23(6), 1078-1086
Surface Properties of Oxidized and Aminated Multi-Walled Carbon Nanotubes
Wellington M. Silva; Hélio Ribeiro; Luciana M. Seara; Hállen D. R. Calado; André S. Ferlauto; Roberto M. Paniago; Cristiano F. Leite; Glaura G. Silva
How to cite this article
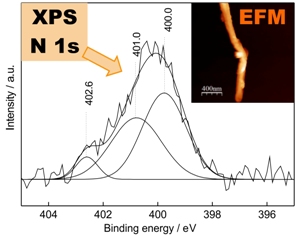
In order to characterize the chemical modifications that were introduced onto the surface of multi-walled carbon nanotubes, studies involving Fourier transform infrared spectroscopy, Raman spectroscopy, thermogravimetric analysis, X-ray photoelectron spectroscopy, transmission electron microscopy and electric force microscopy were performed
https://dx.doi.org/10.1590/S0103-50532012000600012
J. Braz. Chem. Soc. 2012, 23(6), 1087-1097
ESI-MS Study of Speciation in Hydrolyzed Aluminum Chloride Solutions
Mirjana Cvijović; Vesna Kilibarda; Milena Jelikić-Stankov; Ivan Lazarević; Ivan Jakovljević; Ljubinka Joksović; Predrag Đurđević
How to cite this article
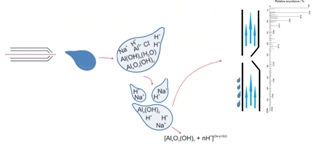
In a chloride-containing medium, aluminum ions form a variety of low and high molecular weight hydrolytic complexes with +1 and +2 charge at pH values lower than 6.0. The complexes may contain coordinated water molecules and chloride ions. The best correspondence between 27Al nuclear magnetic resonance (NMR), potentiometric and mass spectral data is achieved at lower fragmentation voltage and with quadrupole detection
https://dx.doi.org/10.1590/S0103-50532012000600013
J. Braz. Chem. Soc. 2012, 23(6), 1098-1103
Determination of the Hydrophobic Fraction of Ca, Fe, Mg and Zn in Dark Color Honeys Using Solid Phase Extraction and Flame Atomic Absorption Spectrometry
Pawel Pohl; Helena Stecka; Krzysztof Greda; Piotr Jamroz
How to cite this article

Silica gel with bonded phenyl active groups was used to separate the hydrophobic fraction of Ca, Fe, Mg and Zn in dark honeys. Fe was found to be associated with this fraction to the highest degree (7-43% of its total). A strong positive correlation was established between the concentration of Fe in the hydrophobic fraction and the optical density of dark honeys
https://dx.doi.org/10.1590/S0103-50532012000600014
J. Braz. Chem. Soc. 2012, 23(6), 1104-1113
Ab Initio Correlated All Electron Dirac-Fock Calculations for Eka-Francium Fluoride (E119F)
Patrícia S. Miranda; Anna Paula S. Mendes; Jose S. Gomes; Claudio N. Alves; Aguinaldo R. de Souza; Julio R. Sambrano; Ricardo Gargano; Luiz Guilherme M. de Macedo
How to cite this article
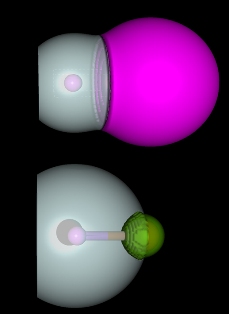
Bader volumes obtained with contour density of 0.001 a.u. at 4-component Dirac-Fock level suggest that eka-francium fluoride (top molecule, E119 in pink color) should be less ionic than lighter alkaline fluoride homologue such as KF (K in green), in contrast to the common chemical belief based on periodic trends - it would be expected the most ionic bond possible. This result should be understood as a consequence of relativistic effects
https://dx.doi.org/10.1590/S0103-50532012000600015
J. Braz. Chem. Soc. 2012, 23(6), 1114-1118
Purification of a Synthetic Pterocarpanquinone by Countercurrent Chromatography
Fernanda das Neves Costa; Alcides José M. da Silva; Chaquip Daher Netto; Jorge L. de Oliveira Domingos; Paulo Roberto R. Costa; Gilda G. Leitão
How to cite this article
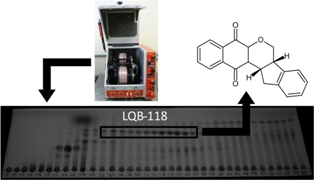
Countercurrent chromatography (CCC) was employed as a useful, fast and economic alternative to conventional chromatography techniques for the purification of a synthetic pterocarpanquinone, LQB-118
https://dx.doi.org/10.1590/S0103-50532012000600016
J. Braz. Chem. Soc. 2012, 23(6), 1119-1123
Efficient Synthesis of Functionalized 1,2,3-Triazoles by Catalyst-Free 1,3-Dipolar Cycloaddition of Nitroalkenes with Sodium Azide
Ting Wang; Xiao-Chun Hu; Xu-Jiao Huang; Xin-Sheng Li; Jian-Wu Xie
How to cite this article
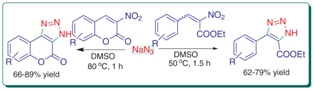
A simple and efficient protocol has been developed for the synthesis of 1,2,3-triazole derivatives by catalyst-free 1,3-dipolar cycloaddition of nitroalkenes with sodium azide under mild conditions. The reaction's scope proved to be quite broad and 1,2,3-triazole derivatives were obtained in good yields
https://dx.doi.org/10.1590/S0103-50532012000600017
J. Braz. Chem. Soc. 2012, 23(6), 1124-1130
Electrochemical Detection of Pb and Cd in Paper-Based Microfluidic Devices
Jianjun Shi; Fan Tang; Honglong Xing; Huxiang Zheng; Lianhua Bi; Wei Wang
How to cite this article
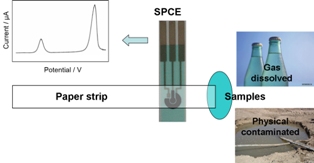
Filter paper strip is combined with screen-printed carbon electrodes (SPCE) to assemble a microfluidic device for detecting Pb(II) and Cd(II) in aqueous samples. The portable microfluidic devices are demonstrated for direct quantification of Pb(II) and Cd(II) in aqueous samples without pretreatment of contaminated samples
https://dx.doi.org/10.1590/S0103-50532012000600018
J. Braz. Chem. Soc. 2012, 23(6), 1131-1139
Electrochemical Reduction of the Mycotoxin Citrinin at Bare and Modified with Multi-Walled Carbon Nanotubes Glassy Carbon Electrodes in a Non-Aqueous Reaction Medium
Vanessa G. L. Zachetti; Adrian M. Granero; Sebastian N. Robledo; María A. Zon; Carlos A. Da Rocha Rosa; Héctor Fernández
How to cite this article
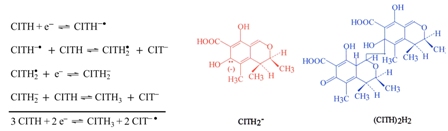
In this study, the electrochemical reduction mechanism of citrinin (CITH) mycotoxin in a non-aqueous reaction medium was investigated. (CITH)2H2 is the dimeric product obtained from the Michael-type nucleophilic addition reaction of CITH2- carbanion with another CITH molecule
https://dx.doi.org/10.1590/S0103-50532012000600019
J. Braz. Chem. Soc. 2012, 23(6), 1140-1145
High-Time Resolution Measurements of Black Carbon Particles in the Exhaust Emissions of a Diesel Engine during Acceleration, Deceleration and Cruise Conditions
Antonio H. Miguel; Anthony D. A. Hansen
How to cite this article
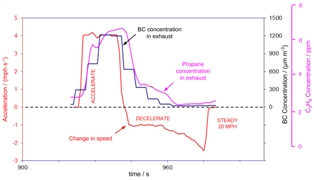
We used an Aethalometer® to measure BC concentrations with 5 s time resolution in the exhaust of a 1995 Dodge RAM 2500 diesel pick-up truck, equipped with an oxidation catalyst, using a chassis dynamometer during the acceleration, deceleration, and cruise modes of the hot start of the MEC01 test cycle (version 7.0)
https://dx.doi.org/10.1590/S0103-50532012000600020
J. Braz. Chem. Soc. 2012, 23(6), 1146-1153
PtSnIr/C Anode Electrocatalysts: Promoting Effect in Direct Ethanol Fuel Cells
Júlio C. M. Silva; Rodrigo F. B. De Souza; Mayara A. Romano; Melina D'Villa-Silva; Marcelo L. Calegaro; Peter Hammer; Almir O. Neto; Mauro C. Santos
How to cite this article
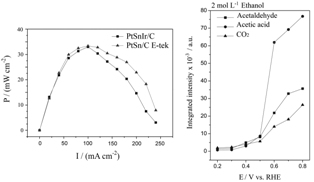
The activities of PtSnIr/C and PtSn/C E-tek electrocatalysts were evaluated in a direct ethanol fuel cell (DEFC) and similar performance was obtained using PtSnIr/C, with half content of platinum. In situ Fourier transform infrared (FTIR) analysis revealed acetic acid as main product generated from ethanol oxidation on PtSnIr/C
https://dx.doi.org/10.1590/S0103-50532012000600021
J. Braz. Chem. Soc. 2012, 23(6), 1154-1165
Determination of Carbamates in Beverages by Liquid-Liquid Extraction with Low Temperature Partitioning and Liquid Chromatography
Simone M. Goulart; Renata D. Alves; Washington X. de Paula; José Humberto de Queiroz; Antônio A. Neves; Maria Eliana L. R. de Queiroz
How to cite this article
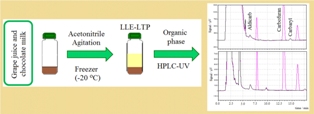
Liquid-liquid extraction with low temperature partitioning was proposed to determine the carbamates pesticides, aldicarb, carbofuran and carbaryl, in grape juice and chocolate milk beverages. The method was quick, cheap and may be easily implemented for routine analysis
https://dx.doi.org/10.1590/S0103-50532012000600022
J. Braz. Chem. Soc. 2012, 23(6), 1166-1173
Determination of Copper in Human Hair and Tea Samples after Dispersive Liquid‑Liquid Microextraction Based on Solidification of Floating Organic Drop (DLLME-SFO)
Soleiman Bahar; Razieh Zakerian
How to cite this article
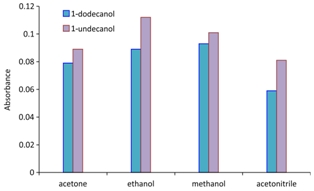
The Figure shows efficiency of DLLME-SFO technique for the extraction of the copper ion with the oxine ligand and using different extraction (1-dodecanol, 1-undecanol) and disperser (methanol, ethanol, acetone, acetonitrile) solvents
https://dx.doi.org/10.1590/S0103-50532012000600023
J. Braz. Chem. Soc. 2012, 23(6), 1174-1185
Oncocalyxone A: Electrochemical, Spectroscopic Investigation and Studies of Its Interaction with DNA, Nucleobases and N-Acetylcysteine
Cicero de O. Costa; Erivaldo de O. Costa; Fabrícia da R. Ferreira; Luciana da S. Viana; Leonardo V. da Silva; Francisco de A. dos Santos Silva; Fabiane C. de Abreu; Isis M. Figueiredo; Waldomiro Pinho Jr.; Edson de S. Bento; Telma L. G. Lemos; Otília D. L. Pessoa; Benjamin Aguilera-Venegas; Ester Norambuena; Claudio Olea-Azar; Marília O. F. Goulart
How to cite this article
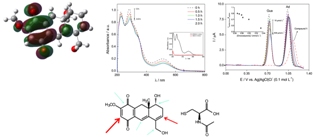
Oncocalyxone A has been shown to be cytotoxic to human tumor cells and DNA reactive. Electrochemical-electron spin resonance revealed the stability of its semiquinone. Experiments with nucleobases and ssDNA (single-stranded DNA) proved the DNA interaction. Nuclear magnetic resonance (NMR) experiments with N-acetylcysteine evidenced its Michael acceptor nature
https://dx.doi.org/10.1590/S0103-50532012000600024
J. Braz. Chem. Soc. 2012, 23(6), 1186-1192
Preparation of Ordered Sulfonated Mesoporous Polymer (OMP-TsOH) from p-Toluenesulfonic Acid and Application in Esterification Reaction of Fatty Acids
Arash Shokrolahi; Abbas Zali; Hamid Reza Pouretedal; Ali Mousaviazar
How to cite this article
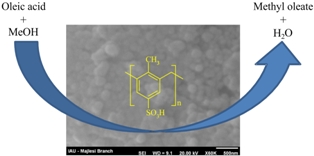
Sulfonated ordered mesoporous polymers were prepared by co-mixing p-formaldehyde, p-toluenesulfonic acid (p-TsOH), sulfuric acid and mesoporous silica (SBA-15 or MCM-48) as a template. The prepared materials were applied as catalyst in oleic acid esterification with methanol
https://dx.doi.org/10.1590/S0103-50532012000600025
Short Reports J. Braz. Chem. Soc. 2012, 23(6), 1193-1198
Fluoride Determination in Carbon Nanotubes by Ion Selective Electrode
Fabiane G. Antes; Juliana S. F. Pereira; Laurien C. Spadoa; Edson I. Muller; Erico M. M. Flores; Valderi L. Dressler
How to cite this article
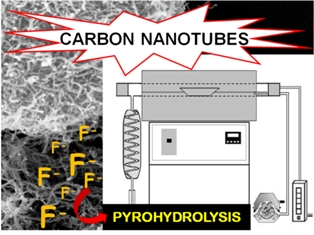
Fluoride determination in single and multi-walled carbon nanotubes was performed by ion-selective electrode after pyrohydrolysis. It was possible to digest up to 1 g of sample and to achieve low LOD
https://dx.doi.org/10.1590/S0103-50532012000600026
Online version ISSN 1678-4790 Printed version ISSN 0103-5053
Journal of the Brazilian Chemical Society
JBCS Editorial and Publishing Office
University of Campinas - UNICAMP
13083-970 Campinas-SP, Brazil
Free access










